Ibogaine & The Heart
Ibogaine is known to effect the heart and not interact well with some other drugs or pre-exisiting conditions. In this article we look at the study ” The Anti-Addiction Drug Ibogaine and the Heart: A Delicate Relation” by Xaver Koenig and Karlheinz Hilber from the Department of Neurophysiology and Neuropharmacology, Center for Physiology and Pharmacology, Medical University of Vienna.
This is a study of the most common causes of death in relation to ibogaine and discuss how they can be avoided.
Chart Showing the details of people who died following ibogaine treatment:
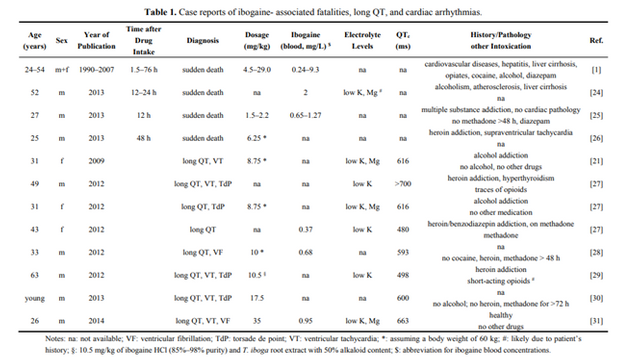
All subjects took ibogaine for treatment of their alcohol or heroin addiction except for one individual seeking spiritual experience.
Between the years 1990 and 2008 a total of 19 fatalities associated with the ingestion of ibogaine were reported from which six subjects died of acute heart failure or cardic arrest.
Summary of most common deaths:
This study found the following:
- Most had Low Magnesium (Mg)
- All had low Potassium (K) – where the data was available.
- 8 cases had Prolonged QT’s
- 2 cases of VF: ventricular fibrillation;
- 5 cases of Torsades de Pointes TdP
- 5 cases of VT: ventricular tachycardia; Were also common alongside the prolonged QT’s
- 4 cases of Sudden death
- In 14 of these cases adequate post-mortem data existed which demonstrated that almost all of the decedents suffered from pre-existing medical conditions or were compromised by the additional intake of other drugs such as opiates and cocaine
- Co-exisiting problems to do with the heart included: coronary artery sclerosis, hypertension, myocardial infarct, cardiac hypertrophy, and dilated cardiomyopathy
- Also some co-exisiting problems regarding the liver are mentioned such as hepatitis, liver cirrhosis and steatosis.
- No signs of neurotoxicity were found in these autopsies, as opposed to the results from earlier experiments on animals.
QT Prolongation and Ibogaine
The QT interval is a measurement made on an electrocardiogram used to assess some of the electrical properties of the heart. It is calculated as the time from the start of the Q wave to the end of the T wave, and approximates to the time taken from when the cardiac ventricles start to contract to when they finish relaxing. In short its a way to monitor your hearts activity and health.
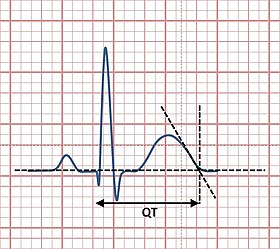
The picture above shows a normal QT, a prolonged QT would look as if it was “stretched out” when this happens there is a risk that the various parts of the heart may not fire in the right order, which can cause BIG PROBLEMS. See below:
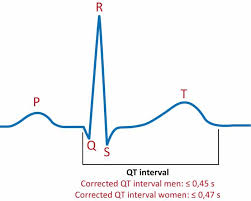
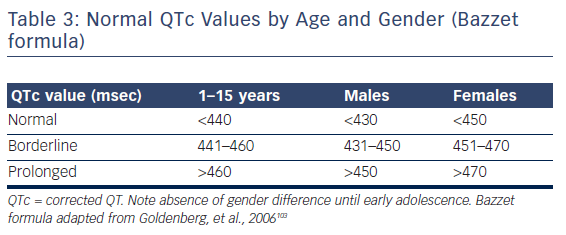
A QTc of >470 and >450 ms was associated with an increased risk of sudden death, for women and men, respectively, and a QT or QTc interval exceeding 500 ms can be regarded a significant risk for torsade de point. At least eight of such cases have been described since 2008.
- QTc values ranged from minor prolongation of only 480 ms to a dramatic one with more than 700 ms in duration.
- Except for one case the prolongation of the QT interval subsequently evolved into ventricular tachycardia (VT) and/or ventricular fibrillation (VF). In many cases this developed into torsade de point (TdP) arrhythmias was observed.
- Only in one case the lack of arrhythmia occurrence was reported. However, in this case the QT interval was only minimally prolonged (480 ms).
In all instances proper medical interventions could’ve saved the lives of these people.
When does it become dangerous?
The ibogaine-ascribed deaths occurred between 1.5–76 after drug intake. But ibogaine only has a half-life of only 4–7 hours so fatalities 24–76 hours after ingesting ibogaine cannot be attributed soley to the alkaloid. Also, QT interval prolongation after ibogaine administration typically lasts for more than 24 hours and has even been observed to occasionally last for longer than a week. Because of that it seems likely that noribogaine (which ibogaine converts to) constitutes the major cardiac risk rather than ibogaine itself.
Risks
Ibogaine should be administered under strict medical observation and continuous electrocardiographic monitoring for an extended time period, which carefully takes noribogaine’s longevity in human plasma into account.
Prior to administration you should carefully consider additional risk factors for drug induced TdP arrhythmias in a patient. These include:
- Female gender
- A prolonged QT
- Bradycardia
- Abnormal electrolyte levels (in particular Low Potassium and Magnesium)
- Pre-existing heart disease
- Ion channel (e.g., hERG) mutations
- Drug-drug interactions
- Genetic variants influencing drug metabolism
For anti-addiction treatment with ibogaine, two among the named risk factors deserve special attention:
- Bradycardia (slow heart rate) Ibogaine itself induces bradycardia
- Hypokalaemia (Low Potassium). Hypokalaemia is frequent in drug users
In Summary
When related to the large number of people who have received ibogaine treatments over many years worldwide, comparably few fatality cases have occurred or have been reported. In addition, drug safety studies on human addicts performed under well-controlled conditions revealed no significant adverse effects.
Baseline screening must include:
- A medical evaluation
- Physical examination
- ECG recording,
- Blood chemistries & electrolyte levels
- Haematological workup
- Psychiatric Report
- Chemical dependency evaluations.
In some cases, more extensive evaluations are required to rule out cardiac risk factors
Cardiac monitoring demonstrated that no electrocardiographic abnormalities were produced or exaggerated following ibogaine administration in subjects that did not have any pre-existing cardiovascular risk factors.
Therefore it appears that ibogaine is well tolerated in drug-dependent subjects and implies that it is not ibogaine alone which causes these problems, but rather the drug in connection with one or more additional risk factors.
More Details on the Risk Factors:
Altered Electrolyte Levels:
Hypokalemia (too little potassium) and often Hypomagnesemia (too little magnesium) are mentioned in case reports of ibogaine associated fatalities and cardiac arrhythmias (changes in heart rate).
- Hypokalemia was detectable when tested. Potassium levels, normally around 3.5–5 mM, were found as low as 2 mM, which reflects a severe hypokalemic condition – this can be attributed to poor diet of drug and alcohol users. The fact that 100% of cases reported this (where information was available) is striking and suggests that this condition predisposes for ibogaine cardiotoxicity.
- Hypokalemia has a two-fold implication in generating long QT and an increased risk of TdP. Firstly it directly decreases hERG currents in the heart. Secondly it can also increase the blockade of hERG currents by drugs .
- Hypomagnesemia was present in 50% of the case reports, and was always accompanied by hypokalemia. It is believed that also hypomagnesemia increases TdP risk
Drug-Drug Interactions and Genetic Polymorphisms
Addicts often have a long history of substance abuse (both illicit and prescribed) and this population is at high risk for adverse drug interactions.
- Concerning the heart, several substances of abuse have been associated with hERG channel inhibition and/or QT interval prolongation. Especially alcohol, cocaine, and methadone. If relevant concentrations of such substances are still residing in the plasma when ibogaine is applied, the drug’s QT prolonging effect, and thus the risk of TdP arrhythmias will be raised.
- Methadone deserves special attention because it additionally has an exceptionally long plasma half- life (15–55 h)
- Another possibility for drug-drug interactions to influence the cardiotoxic potential of ibogaine is impairment of the drug’s metabolism. Ibogaine is metabolised to noribogaine mainly by the enzyme CYP2D6 (30% of most drugs are). Methadone inhibits CYP2D6 so it seems likely that this will impair the pharmacokinetics of ibogaine.
- Since ibogaine’s active metabolite noribogaine has very similar cardiotoxic potential, drugs affecting noribogaine metabolism will be of relevance. Here speculations about specific drug-drug interactions are impossible because noribogaine’s metabolism is basically unsettled but cardiotoxic effects may even persist for weeks after intake of a single dose of ibogaine.
Genetic Differences
Genetic polymorphisms (variants) within the CYP2D6 gene also need to be considered. People can be divided into different classes of metabolisers:
- Ultra-rapid
- Extensive
- Intermediate
- Poor
In poor noribogaine metabolizers, a single ibogaine dose may be enough to induce cardiotoxicity persisting for weeks.
Conclusions
- Ibogaine affects the heart and the cardiovascular system.
- Lowers the heart rate
- It interacts with cardiac ion channels, and these effects most likely determine ibogaine’s potentially life-threatening cardiotoxicity.
Series of Events in Cardiac Events
Based on studies the deleterious events regarding the cardiotoxicity associated with ibogaine appear to happen in the following order:
(1) blockade of repolarizing hERG potassium channels;
(2) retardation of the repolarization phase of the ventricular AP; and
(3) concomitant prolongation of the QT interval in the ECG, ultimately paving the way for life-threatening TdP arrhythmias.
Due to the longevity of noribogaine—ibogaine’s active metabolite—in human plasma, cardiac adverse events may also occur several days, in some cases weeks after intake of a single dose of ibogaine. Noribogaine is believed to be important regarding the long-lasting anti-addictive effect of the treatment.
The use of a drug known to prolong the QT interval must be based on risk-benefit analysis in individual patients. Where benefit outweighs risk, QT prolongation should not limit necessary therapy.
This information was mostly gleaned from: The Anti-Addiction Drug Ibogaine and the Heart: A Delicate Relation. Which can be found here:
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4382526/pdf/molecules-20-02208.pdf

Most of this website content was written by Rich Hughes, as well as the website creation itself.
In the photo to the left, he is the man on the right hand side.
He worked and volunteered for more than 5 years at various ibogaine detox clinics in México and Portugal before striking out on his own in Spain, his clinic is called Madera Sagrada which is Spanish for sacred wood, the indigenous Bwiti name for iboga.
During this time he has experienced the good, the bad and the ugly of ibogaine treatment; from the countless dozens who gained their freedom and even witnessing a death whilst working as a volunteer. This drove him to better understand the health and safety aspects, including how to read an ECG/EKG and an advanced first aid course.
It is with his hard won information, and a desire to see ibogaine knowledge distributed around the world, saving lives, as it has saved his life from Crack and heroin addiction.
"I´m just a common man who through his own experience and desire has studied ibogaine and iboga and their correct administration. I stand on the shoulders of giants and through my web and graphic design skills I´ve decided to disseminate this info to a wider audience, with the hope of spreading liberty to those that desire it
www.maderasagrada.org
maderasagrada@protonmail.com
+34 643280060 (Whatsapp & Telegram available)
Learn about the jaw-dropping benefits of Ibogaine and Iboga. They do so much more than just addiction treatment
Ibogaine can be dangerous for some people. Educate yourself to see if you are a good candidate for this work
Preparing for your iboga ceremony or ibogaine treatment. Mentally, emotionally, physically and spiritually.
Learn what medication and medical health conditions are a bad mix with ibogaine and iboga.
Make sure you go to a good place! There are some shady operations going on out there…
Ibogaine and the heart have a delicate relationship. Indepth article about the action on the heart.
Talk to us!
Our knowledgeable volunteers will share their personal experience of ibogaine with you and advise you on how to find a good clinic.
+34 643280060
For a free call back:
Text, Whatsapp, Telegram app or email us!
brain@ibogainehotline.co
Or by clicking the blue speech bubble to the right of the screen to chat to us right now!
Join our facebook group and get first hand advice from people who have already made their journies with ibogaine www.facebook.com/groups/1521724148001426/
Like our Facebook page www.facebook.com/ibogainehotline

We wish you luck on your journey. This incredible medicine has changed so many lives, but to do safe and effective work it must be treated with respect.
It’s important that you find a good provider or clinic to do your ibogaine treatment with. Don’t end up with the cowboys!
We do our best to keep this website updated as our collective knowledge and experience of the use of ibogaine grows, and we would love you contributions and suggestions. Email us






